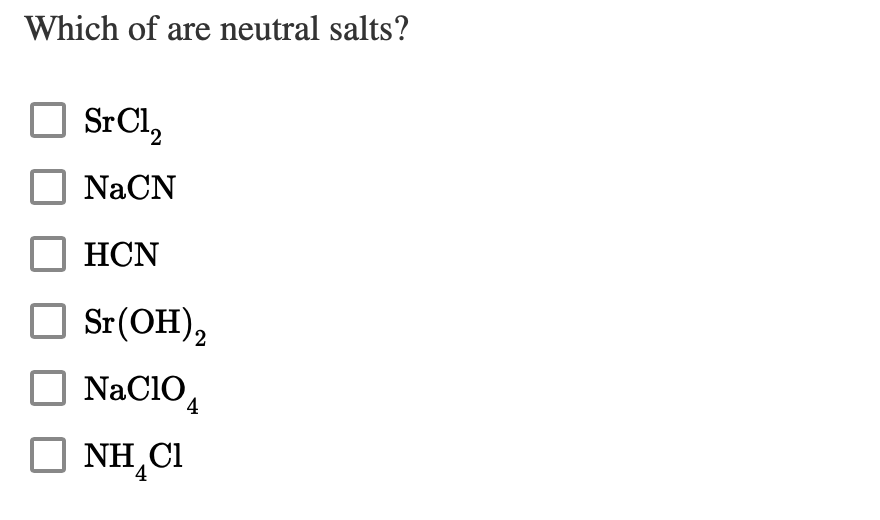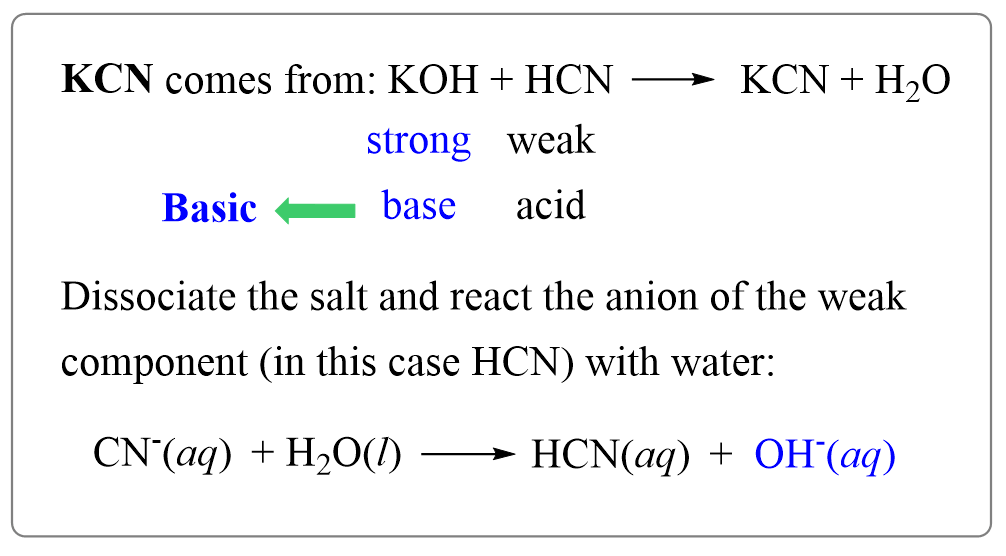
SOLVED: The weak acid hydrocyanic acid, HCN, and the strong base sodium hydroxide react to form the salt sodium cyanide, NaCN. Given that the value of Ka for hydrocyanic acid is 4.90×10−10,

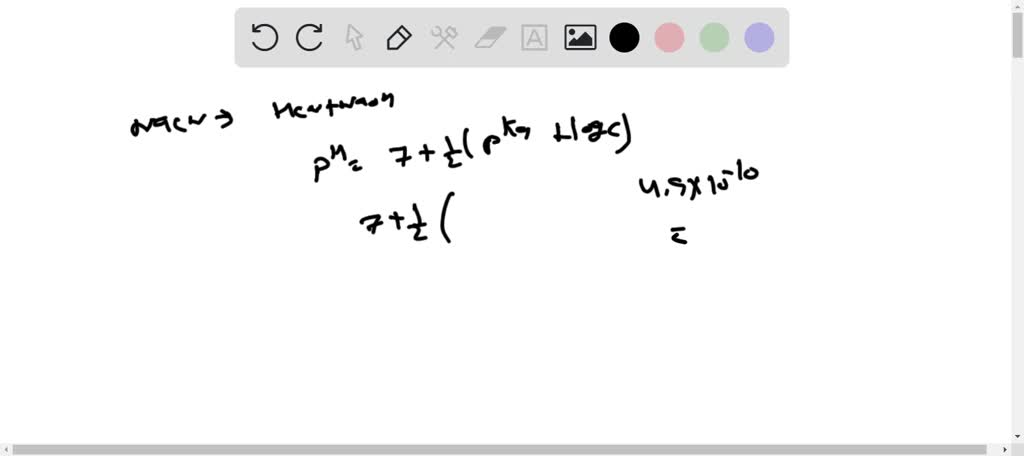
Ammonium cyanide is a salt of NH(4)OH(K(b)=1.8xx10^(-5)) and HCN (K(a)=4.0xx10^(-10)). The hydrolysis constant of 0.1 M NH(4)CN at 25^(@)C is :

SOLVED: idantify salt solution KCN NHg NHA CN givan: Kb NH3 HCN neutral slightly basic slightly acidic can not toll 1,8 x 10-5 49* 10F1

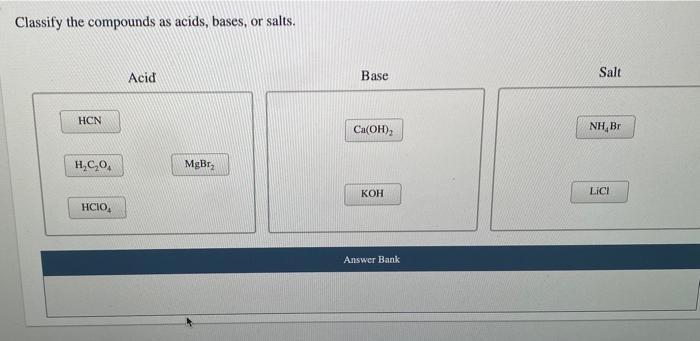
1 Acid-Base Properties of a Salt Solution One of the successes of the Brønsted- Lowry concept of acids and bases was in pointing out that some ions can. - ppt download

SOLVED:Sodium cyanide is the salt of the weak acid HCN. Calculate the concentration of H3 O^+, OH^-, HCN, and Na^+ in a solution prepared by dissolving 10.8 g of NaCN in enough

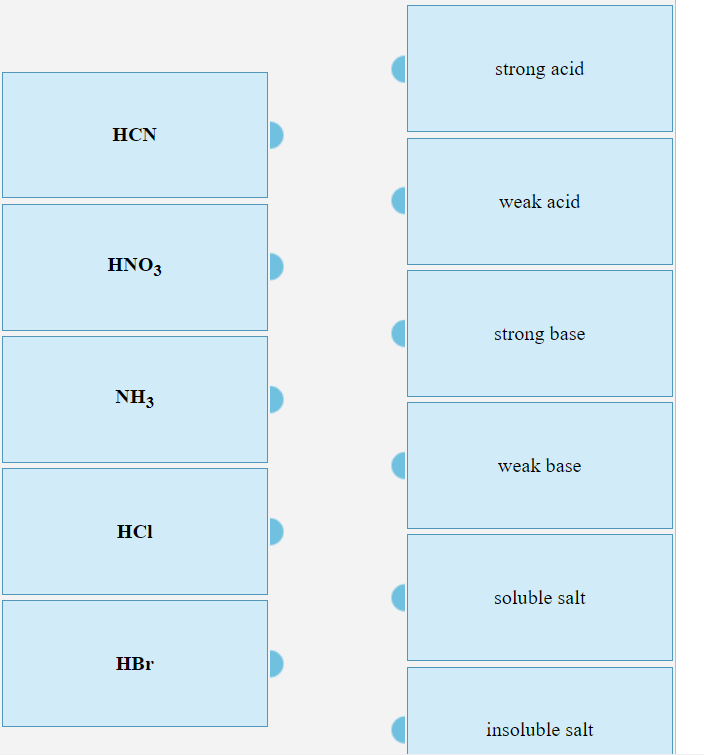
Acid-base Equilibria n K a and K b n % dissociation of weak acid/bases n pH of weak acid/base solutions n pH of salt solutions n Buffers n pH of buffer. -

Hydrogen cyanide (HCN)- Lewis acid Structure, Molecular mass, Physical and Chemical Properties, Uses with FAQs of Hydrogen Cyanide.
![Salts of HCN‐Cyanide Aggregates: [CN(HCN)2]− and [CN(HCN)3]− - Bläsing - 2020 - Angewandte Chemie International Edition - Wiley Online Library Salts of HCN‐Cyanide Aggregates: [CN(HCN)2]− and [CN(HCN)3]− - Bläsing - 2020 - Angewandte Chemie International Edition - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/ec0eebe8-512c-4b8b-a209-39a640a4e6d1/anie201915206-toc-0001-m.jpg)
Salts of HCN‐Cyanide Aggregates: [CN(HCN)2]− and [CN(HCN)3]− - Bläsing - 2020 - Angewandte Chemie International Edition - Wiley Online Library

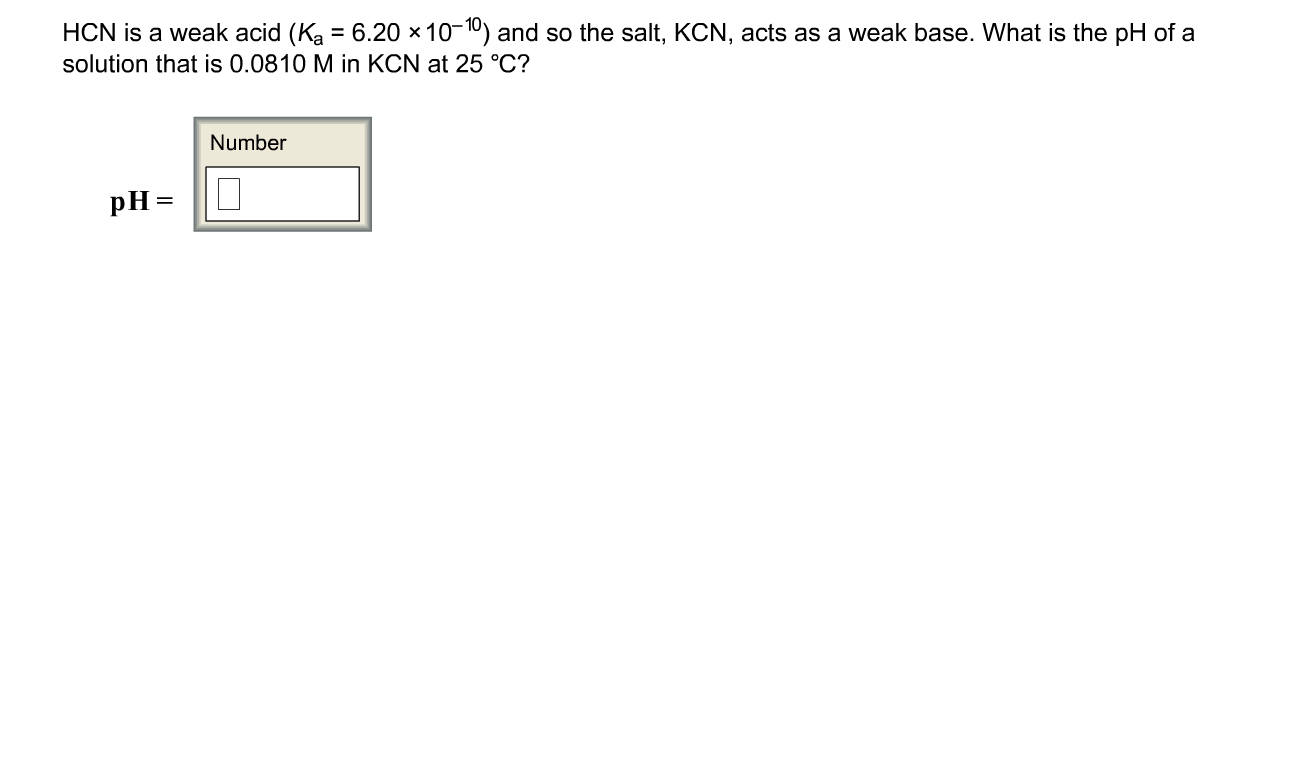
SOLVED: HCN is a weak acid (Ka 6.20 x 10-10), so the salt; KCN acts aS a weak base. What is the pH of a solution that is 0.0330 M in KCN

Hcn 0227 Series Skid Steer Loader Front End Salt Spreader Attachment - Buy Self-loading Sand Spreader,Tractor Attachment Salt Spreader 022704,Liugong Clg777 Hot Sale Chinese Sam388 Backhoe Loader Product on Alibaba.com